R-107
Claritas is developing a novel treatment for COVID-19 and Other Viral Infections
R-107 is Claritas’ patented nitric oxide-releasing compound. Nitric oxide is the immune system’s first line of defense against viral infection. When a viral threat is present, white blood cells migrate to the area of infection and release a burst of nitric oxide that crosses into the infected cell to inactivate the virus. R-107 is designed to augment the body’s natural release of nitric oxide to significantly hasten and strengthen the body’s ability to eliminate the virus.
There is an extensive body of in vitro and in vivo data demonstrating the ability of nitric oxide to prevent the replication and transmission of various RNA viruses, such as those that cause COVID-19 infection, as well as viruses that cause the influenza and the common cold.
Unlike nitric oxide, which is a gas that must be administered by a trained respiratory therapist using special delivery equipment, R-107 is a liquid that may be easily administered orally in a capsule, or nasally though use of a spray, or by intramuscular injection. Due to its ease of administration, R-107 may overcome the challenges inherent in administration of nitric oxide gas, and transform nitric oxide therapy into a potentially best-of-care treatment for COVID-19 infection, and potentially other viral infections.
 Technology
Technology
R-107 is intended as the first therapy that simultaneously targets coronaviral disease via four independent mechanisms of action:
- Eliminates coronaviral replication
- Thwarts coronaviral attachment to the epithelial angiotensin-converting enzyme 2 (hACE2) for the coronavirus spike protein
- Protects organs from the oxidizing and nitrosating effects of cytokine-induced generation of peroxynitrite (ONOO¯)
- Relieves pulmonary hypertension, a major driver of acute respiratory failure
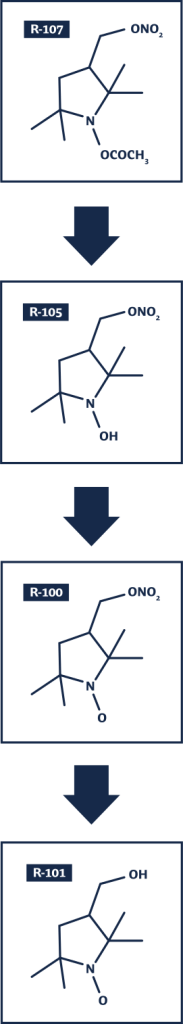
As shown in the schematic, R-107 is hydrolyzed to form the intermediate R-105 (3-nitratoproxyl-hydroxylamine). R-105 subsequently oxidizes to form R-100, the active payload molecule serving as: (a) a nitric oxide (“NO”) donor via its organic nitrate (to form the denitrated entity hydroxymethylproxyl (R-101), and (b) a broad-spectrum catalyst of reactive oxygen species (“ROS”) degradation via its nitroxide moiety.
The free radical catalytic domain acts as a superoxide dismutase mimetic, a catalase mimetic, and a peroxynitrite degradation catalyst. R-107 is a stable non-viscous liquid that is readily administered as a neat entity once daily by intramuscular injection or via an oral route, yielding sustained systemic drug levels in rat, dog, and sheep. R-107 is intended as a donor of NO that does not develop nitrate tolerance and is thus amenable to a 1-2 week course of in-patient daily administration for treatment of life-threatening disease.
Current Development Program
Claritas is working with Salzman Group to develop R-107 for the treatment of COVID-19 and other viral infections. Salzman Group has received $25M in federal funding to date for advancement of R-107 and R-100 for treatment of chlorine inhalational lung injury (“CILI”) and sepsis.
The government does not enjoy any commercial rights to the technology. The contract is part of efforts by ASPR’s Biomedical Advanced Research and Development Authority (“BARDA”) to develop medical products and procedures to protect health and save lives in a terrorist attack, including those using toxic industrial chemicals such as chlorine. The funding supported the advancement of R-107 in manufacturing, pharmacology, and toxicology.
(“BARDA”) to develop medical products and procedures to protect health and save lives in a terrorist attack, including those using toxic industrial chemicals such as chlorine. The funding supported the advancement of R-107 in manufacturing, pharmacology, and toxicology.
Biological Targets
R-100, the active payload of R-107, principally interacts with six discrete biological targets that are crucial to the pathophysiology of COVID-19 disease:
Direct inhibition of viral uptake and replication
(#1) Cysteine residue of the spike (S) protein of COVID-19
Intracellular entry by COVID-19 requires post-translational thiol esterification of a critical Cyteine residue of the S protein by the long-chain fatty acid palmitate in order to achieve binding of hACE2 and entry into epithelial cells (lung, enterocyte, renal tubule). NO nitrosylates this Cysteine residue, thereby interfering with its palmitoylation and preventing the binding and entry of Coronavirus (SARS-1 and COVID-19), as reported by the Karolinska institute and KNOW BIO. Exogenous provision of NO via various NO donors or NO gas provides complete blockade of Coronaviral entry. As reported by KNOW BIO, in vitro viral reduction of COVID-19 virus exceeded 99.9% over 24 hours after a single pulse of NO. It is expected that NO blockade of the S protein is refractory to mutation since palmitoylation of the Cysteine residue is obligatory for viral pathogenesis. This approach constitutes an advantage relative to the deployment of therapeutic monoclonal antibodies which are intrinsically restricted to a specific epitope of the S protein and are thus susceptible to viral evasion by mutation which alters the epitope.
(#2) Replication of Coronaviral RNA
In cultured Vero cells, NO donors and NO gas covalently bind and inhibit SARS-CoV-2 3CL, a SARS-CoV-2 protease activity required for viral replication of COVID-19. This is consistent with S-nitrosation of the enzyme active site cysteine (Cys145).
Activation of guanylate cyclase (GC) in the pulmonary vascular smooth muscle
(#3) Nitrosylation of the iron atom in the heme center of guanylate cyclase reduces pulmonary hypertension
COVID-19 pneumonitis, and resultant ARDS, may produce severe pulmonary hypertension, resulting in acute right heart failure, pulmonary shunt, hypoxemia, and respiratory failure requiring intubation and mechanical ventilation. R-107 normalizes pulmonary vascular resistance (PVR) and pulmonary blood pressure in rats with monocrotaline-induced pulmonary hypertension and in sheep with chlorine inhalational lung injury (CILI). The organic nitrate moiety of the active payload R-100 releases NO, presumably in the form of a thionitrate. This intermediate form of NO nitrosylates the iron atom in the heme center of guanylate cyclase, inducing a conformational change in the enzymatic center that activates catalysis of GTP to cGMP. The generation of intracellular cGMP triggers relaxation of the pulmonary smooth muscle. Changes in systemic vascular resistance (SVR) are not observed unless suprapharmacologic doses of R-107 are employed. Hence, R-107 acts as a pulmonary-selective vasodilator.
Redox catalysis
(#4) Dismutation of O2-, (#5) reduction of H2O2, and (#6) catalysis of ONOO–
 Superoxide anion (O2¯) forms abundantly during cytokine storm secondary to activation of infiltrating monocytes and neutrophils, as well as impaired coupling by Complex 1 of the mitochondrial electron transport chain. Superoxide anion so produced reacts immediately (reaction rate < one billionth) with ambient NO to form the potent nitrosating agent peroxynitrite (ONOO¯), hence exogenous therapeutic provision of pure NO gas is a two-edged sword. In contrast, delivery of NO by R-107 does not result in the formation of peroxynitrite, because: a) R-100 removes superoxide directly via catalysis by its nitroxide moiety, thereby blocking the formation of peroxynitrite; and b) R-100 directly degrades peroxynitrite. Accordingly, administration of R-107 does not induce peroxynitrite formation and, in fact, prevents endogenous peroxynitrite formation in endotoxin-challenged mice where ambient superoxide anion is in excess. We expect that COVID-19 pneumonia also is associated with abundant superoxide anion in the lung. Hence, inhaled NO while helpful overall is not the ideal means to deliver NO to the lung of the COVID-19 infected patient.
Superoxide anion (O2¯) forms abundantly during cytokine storm secondary to activation of infiltrating monocytes and neutrophils, as well as impaired coupling by Complex 1 of the mitochondrial electron transport chain. Superoxide anion so produced reacts immediately (reaction rate < one billionth) with ambient NO to form the potent nitrosating agent peroxynitrite (ONOO¯), hence exogenous therapeutic provision of pure NO gas is a two-edged sword. In contrast, delivery of NO by R-107 does not result in the formation of peroxynitrite, because: a) R-100 removes superoxide directly via catalysis by its nitroxide moiety, thereby blocking the formation of peroxynitrite; and b) R-100 directly degrades peroxynitrite. Accordingly, administration of R-107 does not induce peroxynitrite formation and, in fact, prevents endogenous peroxynitrite formation in endotoxin-challenged mice where ambient superoxide anion is in excess. We expect that COVID-19 pneumonia also is associated with abundant superoxide anion in the lung. Hence, inhaled NO while helpful overall is not the ideal means to deliver NO to the lung of the COVID-19 infected patient.
Nitric Oxide
Clinical support for the role of NO in COVID-19 infection
A trial of inhaled NO was effective in treating a small series of patients with SARS-1 pneumonia more than a decade ago. These findings encouraged a recent study at the Mass General Hospital, reported in September 2020, wherein six pregnant patients with severe and critical COVID-19 infection received rescue therapy with inhaled NO. The treatment was well tolerated. By day 22, five of the six patients, who received no other antiviral medication, tested negative twice for COVID-19. All six patients were discharged, and have delivered healthy, COVID-19 free, infants. These data represent a breakthrough in the potential treatment of COVID-19 infection. A dozen trials of inhaled NO are underway currently for amelioration of COVID-19 pneumonia (see Appendix below).
Unique advantages of R-107 vs. inhaled NO Gas
• The gaseous delivery of NO only targets the lung, because it is instantaneously inactivated once it reaches the pulmonary circulation via its nitrosylation of iron in hemoglobin present in circulating erythrocytes. COVID-19 is a systemic infection, with involvement of the gut mucosa, renal tubules, heart, and CNS; hence inhaled NO cannot treat extrapulmonary disease. R-107 delivers R-100 systemically, reaching all organs where infection is active or preventing dissemination of active pulmonary infection to the periphery.
• Inhaled NO only reaches areas of lung with excellent ventilation. Since ARDS is associated with profound ventilation-perfusion mismatch, and many areas of the lung are poorly ventilated, inhaled gaseous delivery of NO misses delivery to the full lung. This is potentially a major limitation of NO gas delivery.
• Inhaled NO gas is expensive to deliver because it necessitates the use of specialized gas delivery systems managed by respiratory therapists and monitored continuously by nursing personnel. R-107 is administered by a daily intramuscular injection. Accordingly, R-107 can be readily delivered in a pandemic setting without complex equipment and labor-intensive set-up and monitoring.
• Gas delivery systems for inhaled NO are not available in sufficient number to handle a pandemic. R-107 dosage (1 mL) is easily produced, distributed, stored, and administered.
• Inhaled NO is a two-edged sword. Useful for all of the above reasons described, but also a reactant to form the toxic species peroxynitrite. R-100 does not form peroxynitrite.
• NO gas has a half-life of 2-3 seconds, and hence must be administered continuously. Loss of NO delivery for even several minutes results in profound and life-threatening rebound pulmonary hypertension. R-100 in contrast has a half-life of 4-8 hours in large animals.
Unique advantages of R-107 vs. existing organic nitrates
• Nitroglycerin has a half-life of minutes and rapidly induces tolerance.
• Isosorbide monitrate and isosorbide dinitrate are relatively weak NO donors, do not have much effect on pulmonary hypertension, and also induce tolerance.
Unique advantages of R-107 vs. PD5 inhibitors and GC activators
• Sildenafil and other PD5 inhibitors (which block degradation of cGMP), and GC activators (which trigger formation of cGMP), do not release NO.
• They may be effective in reducing pulmonary hypertension but they have no antiviral activity.
